Our Research
Electron and Hole Upconversion
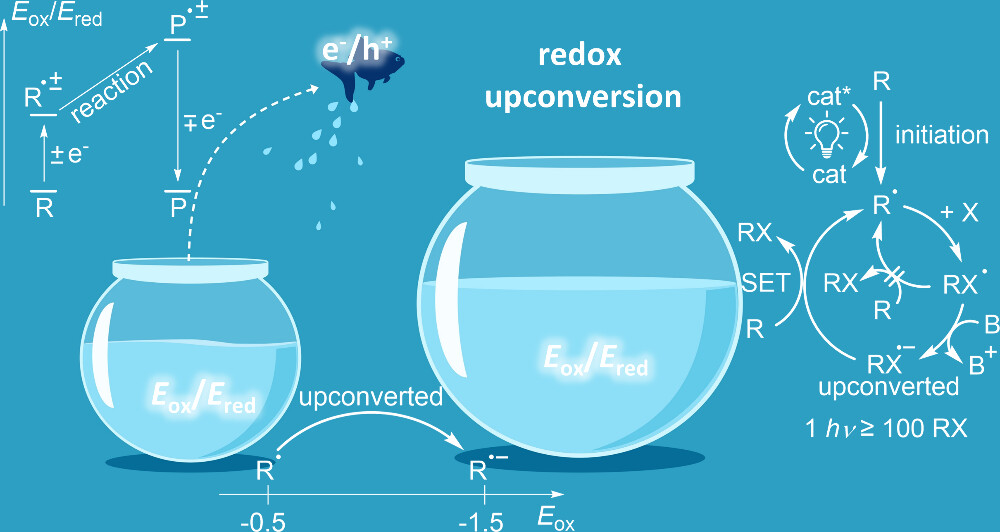
Using an electron as a catalyst is appealing, both conceptually and practically. Not only are electrons cheap and abundant, but they are also traceless and recyclable, so the use of electrons as catalysts is an inherently green chemical strategy. Many chemical transformations are made possible by electron transfer, as illustrated by the remarkable increase in the applications of electrochemical and photoredox methods in chemistry. For example, in several documented cases, a single absorbed photon can lead to the formation of >100 molecules of the product, behavior known to originate from chain processes. We explore the underlying reasons for this efficiency, identify the nature of common catalytic chains, and highlight the differences between HAT and SET chains. Our goal is to illustrate how the phenomenon of redox upconversion serves as a thermodynamic precondition for electron and hole catalysis.
Synthesis of Symmetrically Functionalized Triphenylenes (SFTPs)
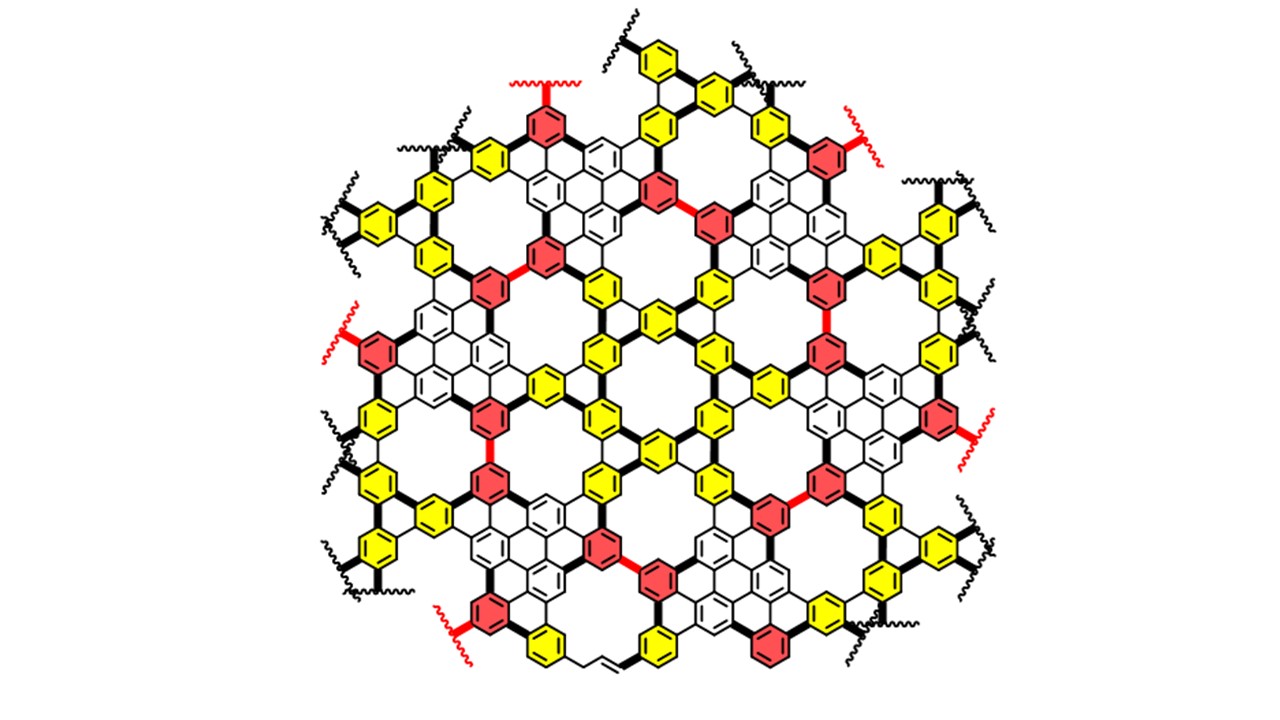
Our intramolecular approach takes advantage of the energy-rich alkyne functionality to transform a simple benzene core via a highly regioselective cyclization into a hexahydrotriphenylene core, followed by aromatization into triphenylene derivatives that are symmetrically decorated with functional substitutents amenable to cross-coupling reactions and other transformations. The resulting SFTPs, with their D3h symmetry, serve as ideal building blocks for the synthesis of two-dimensional graphenic materials, polyhelicenes, and mesogenic molecules, which can be employed in the fabrication of discotic liquid crystals (DLCs).
Design and Synthesis of Organic Materials
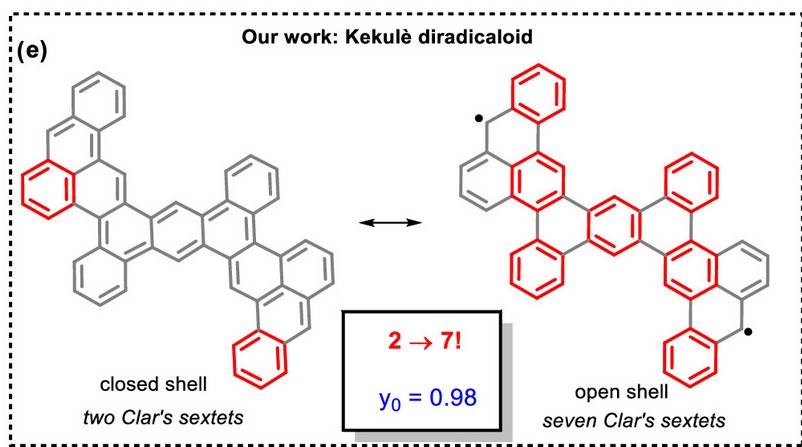
We strategically design, synthesize, and characterize organic diradicaloids and triradicaliods, a class of highly reactive and unstable molecules. Our approach focuses on enhancing stability by leveraging Clar’s sextet rule, incorporating heteroatoms, and conjugated scaffolds for electronic stabilization, and introducing bulky substituents to protect unpaired electrons, thereby preventing dimerization through kinetic stabilization. For example, we take advantage of electron-rich alkynes through peri-annulation reactions to construct carbon-based and nitrogen-doped diradicaloids. To unlock their full potential, we employ advanced characterization techniques, including room- and variable-temperature EPR, SQUID, CV, UV-Vis, NMR, and computations, to probe their unique electronic and spin properties. These extraordinary molecules hold immense promise for spintronics, semiconductors, chemical sensors, detectors, batteries, and magnetic materials. Additionally, we explore porous PAHs by functionalizing pyrene, leading to materials with remarkable photophysical properties.