Our research group delves into the exciting field of self-propelling microstructures, taking cues from the intricate mechanisms behind biological transport and locomotion. Utilizing the principles of chemical gardens, we have successfully developed self-assembling and self-propelling inorganic structures with promising applications in biomedicine, environmental science, and advanced robotics.
A key accomplishment is the creation of hollow manganese oxide tubes derived from self-assembling calcium carbonate structures. These tubes display self-propulsion when placed in H2O2 solutions due to the catalytic production and ejection of oxygen bubbles. We have also observed the collective behavior of these tubes revealing fascinating patterns like cooperativity and swarming dynamics.
Building on this foundation, we have developed micro-submarines that change their depth in response to catalytic gas production. These structures, made of silica-supported CuO and assembled using chemical garden principles, showcase an impressive ability to self-propel. In H2O2 solutions, they produce oxygen gas bubbles that lift the micro-submarine to the air-solution interface before sinking back down to the bottom.
Our group has not only investigated the behavior of these self-propelling structures but also developed an efficient method for producing them. Traditional production methods can be complex and time-consuming, limiting widespread use. Our approach, however, involves injecting metal salts into silicate solutions, creating hollow, cylindrical structures rich in catalytic manganese oxide and featuring an insulating outer layer of inert silica. In dilute H2O2 solutions, these structures propel themselves forward by ejecting streams of oxygen bubbles.
Furthermore, we have explored the possibility of sound measurements by positioning a microphone just above tiny drops of H2O2 solution. This experimental setup allowed us to analyze and characterize the acoustic properties of these self-propelling microstructures, shedding light on the underlying mechanisms and dynamics at play.
For chemical reactions to occur, the reactant molecules should first meet. If the reaction medium is stirred, then this is not a problem, the molecules interact in a homogeneous medium. But in unstirred media, molecules are heterogeneously distributed and their transport occurs via diffusion.
Amazingly, interesting macroscopic patterns can emerge from chemical systems simply by coupling reaction and diffusion. A classic example, is the Belousov-Zhabotinsky reaction which organizes spiral waves in two spatial dimensions and scroll waves in 3D. These rotating structures are essentially oxidation waves that sweep through a chemically reduced medium much like a wildfire propagates across a forest. Understanding these phenomena from an experimental and theoretical perspective is giving novel insights into other excitable media, specifically in biophysical fields such as neurology and cardiology.


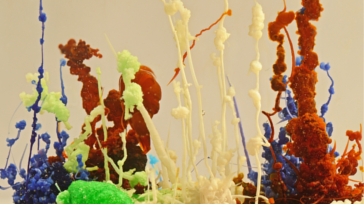

Chemical or silica gardens are tubular precipitation structures that grow during the interaction of metal salt solutions with silicates, carbonates or other selected anions. Their growth characteristics and attractive final shapes derive from a complex interplay between reaction-diffusion processes and self-organization. A classic demonstration experiment in chemistry, the study of chemical gardens gained importance over the years due to their potential to bridge the nanoscopic and macroscopic world, the interesting patterns and motifs that span from them and their association to hydrothermal vents and one hypothesis for the origin of life on Earth. This emerging research topic has been named Chemobrionics.
Our group is actively contributing to the advancement of this field. From establishing new experimental setups for controlled growth of precipitation tubes, analyzing their composition and characteristics up to using these tubes as platforms for quantum dots and other applications, both fundamental knowledge and technology are of interest to us.
Chemobrionics explores the boundaries between chemistry, physics and engineering covering hot topics such as self-assembly and self-organization, hydrodynamics, materials science and characterization. It is challenging, no doubt, but rewards you with a comprehensive education and the thrill of working in a new research field full of opportunities.
An excitable medium is a spatially extended nonlinear system that can propagate waves without damping. These systems are able to self organize into a special vortex pattern rotating around a one-dimensional filament. Often these same patterns are found in the chemical and biological world, such is the case with cardiac arrhythmia.
The dynamics of such a system here is described by mathematical models together with computational simulations, which have been shown to be accurate by agreements with experiments and theory. Recently, we have incorporated the use of high efficient computing techniques, such as parallel computing and GPU computing. In addition, these computational methods help us to understand the dynamics of the system like the evolution of the filament, which is often invisible in experimentation.
The normal heartbeat is mediated by the conduction of electrical waves through cardiac tissues. The activity of these waves indicates the degree of heart’s health and has been clinically utilized in the diagnosis of heart diseases. The waves are measured by a device called the electrocardiogram (ECG). The nature of ECG is that the cardiac tissue is an excitable medium that can response to external stimulus and propagate waves by a diffusion process.
Despite a wealth of experimental data, characterizing rapid electrophysiological changes are complex and challenging. Our goal is to accurately predict the electrophysiology of heart, and thus increase the efficiency of diagnoses while minimizing possible health problems. Our computational methods are designed to: bridge over challenges in modern cardiac medicine, significantly enhance the analysis of the heart, and provides an insightful understanding of cardiac systems and diseases.
Stable Spiral
Unstable Spiral

Can purely inorganic processes form life-like shapes?
Yes!
As we have learned from a unique crystallization system. The co-precipitation of silica and barium carbonate in alkaline media produces life-like structures such as braided worms, twisted helices, and leafy sheets. Known as biomorphs, these crystal aggregates have blurred the boundaries between fossils of animate beings and inorganic products of geochemical processes.
However, the merits of this system are not limited to the aesthetics. At the nano-scale, silica-carbonate biomorphs are composed of crystalline rods aligned within an amorphous matrix to form a hierarchical architecture. This arrangement mimics the assembly of natural biominerals such as bones, teeth, and shells. In fact, even the actual biomorph growth mechanism is not a conventional crystallization and offers an excellent model for studying in vitro biomimetic crystallization. The fundamental insight gained from this representative system will significantly expand the toolbox of crystallization methods and allow the rational design of biomimetic materials with controlled properties.